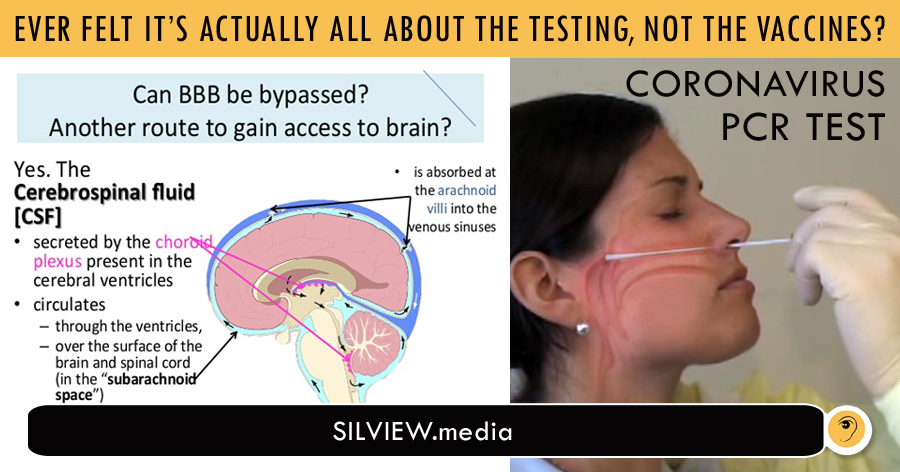
In an increasingly baby-minded world, I had to pull out the crayons again.
I feel embarrassed for the human race that I have to explain this and so many people need to see it.

Is It ‘Eugenics’ to Abort Unborn Babies with Down Syndrome?
By Alexandra DeSanctis, staff writer for National Review and a visiting fellow at the Ethics and Public Policy Center.
“On the legal blog Dorf on Law, in an article by Sherry F. Colb, a Cornell University law professor. Colb argues that, because eugenics is defined as “a movement . . . aimed at improving the genetic composition of the human race,” it is inapt to call selective abortions “eugenic,” because a woman who chooses abortion after a Down-syndrome diagnosis “understands that she is thereby doing virtually nothing to alter the human genome.”
But Colb ignores another meaning of the adjective “eugenic”: “relating to or fitted for the production of good offspring.” Though the term “eugenics” undoubtedly evokes a program of controlled, selective breeding to reshape a population, it is entirely accurate to describe as “eugenic” an individual choice to eliminate a child deemed “unfit,” even in just one instance.
Colb concludes with this argument:
What if everyone pregnant with a DS fetus terminates? What then? Do we want to live in a world in which DS people are extinct? No. There is no question but that people with DS, like people with all sorts of other challenges, enrich our world and teach us to tolerate those who differ from ourselves. It would indeed be sad if the world contained no one with DS. But just because we want a group of people in the world does not entitle us to conscript individuals to create such people in their wombs.
But of course, forbidding abortions chosen on the basis of disability cannot rightly be described as “conscripting individuals to create such people in their wombs.” When a pregnant mother receives a prenatal Down-syndrome diagnosis, she has already created a human being who might have Down syndrome (though such tests have been known to be wrong). Forbidding a woman from actively killing her unborn child based on its disability is not the same thing as conscripting her into creating that child.
That defenders of legal abortion are reduced to such arguments is telling. In the end, it doesn’t matter much whether we can rightly label certain abortions “eugenic” or whether one side of the debate has the most accurate history of racial discrimination and population control.
What matters is that, in Ohio, lawmakers have laid down a marker establishing that it is wrong and therefore that it is now illegal to end the life of an unborn human being simply because he or she is diagnosed with a chromosomal abnormality. Supporters of abortion refuse to respond to this argument, because to do so would expose the logic of all abortion, which, regardless of disabilities, grants some human beings the power to declare the lives of others not worth living.”

Gates conducted an interview with Bill Moyers on PBS to explain the rational for his charitable contributions:
MOYERS: You could have chosen any field, any subject, any issue and poured billions into it and been celebrated. How did you come to this one? To global health?
GATES: The one issue that really grabbed me as urgent were issues related to population… reproductive health.
And maybe the most interesting thing I learned is this thing that’s still surprising when I tell other people which is that, as you improve health in a society, population growth goes down.
You know I thought it was…before I learned about it, I thought it was paradoxical. Well if you improve health, aren’t you just dooming people to deal with such a lack of resources where they won’t be educated or they won’t have enough food? You know, sort of a Malthusian view of what would take place.
And the fact that health leads parents to decide, “okay, we don’t need to have as many children because the chance of having the less children being able to survive to be adults and take care of us, means we don’t have to have 7 or 8 children.” Now that was amazing.
So Gates is interested in improving health because he believes that would reduce the amount of people on the planet. His goal is not to help people but to eliminate them. He states that if people are healthy that they will want fewer kids but he doesn’t offer evidence to support this and frankly it doesn’t appear to make much sense. Why would a sick person who could die at anytime want to have kids if they knew there was a good possibility they wouldn’t be around to support the child? Does Gates really believe this or is this just his cover story so not arouse any suspicions about his true motivations? Gates also admits that he notes that he previously shared the opinion with Malthus that health should not be improved because that would encourage population growth. If you remember Malthus wanted villages built near sewage to encourage disease. Now he doesn’t disagree with Malthus that population growth is bad he only disagrees on how to reduce population.
I don’t believe that Gates’ actually thinks that improving health reduces population. I think that he is using global health as a stalking horse to eliminate population. Gates’ could donate money to provide basic healthcare to poor Africans like Doctors Without Borders, he could build hospitals, and he could help provide low cost health insurance to the millions who can’t afford it. Bill Gates money could be spent improving access to safe drinking water and providing sanitation services. His money is spent on any of this noble The elites of the world choose to spend the tax dollars of the American middle class on contraceptives, abortions, and vaccines. Kenyan gynecologist Dr. Stephen Karanja observed, “USAID and other Non-Governmental organizations funded mainly by the U.S. Government have targeted our people with a ruthlessness that makes one shudder. Our health sector has collapsed. Thousands of the Kenyan people will die of malaria, whose treatment costs a few cents, in health facilities whose shelves are stocked to the roof with millions of dollars worth of pills, IUDs, Norplant, Depo-Provera, most of which are supplied with American money.”
“Many are maimed for life. The hypertension, blood clots, heart failure, liver pathology and menstrual disorders cannot be treated due to the poor health services…. Malaria is epidemic in Kenya. Mothers die from this disease every day because there is no chloroquine, when instead we have huge stockpiles of contraceptives.” – SOURCE – I used this not for authoritativeness, but for logic and because it very much speaks my mind too. And I fact-checked it.

SOURCE
THE AFRICAN PERSPECTIVE ON IT
Population Control is GENOCIDE
(This interview with Sister Aset was first published in Global Africa Pocket News (GAP News) Vol. 1, No. 7 Sept. 1994. It was submitted to Caribbean Times in January ’96 but never published) #14
SOURCE
What is population control?
The United Nations Population Fund would like us to believe that it is a benign process of ‘voluntary’ application of ‘family planning’ to control the ‘rate of growth’ of the world’s ‘sustainable’ population within ‘manageable’ levels in relation to the amount of ‘food’ and ‘consumable goods’ the earth can produce. That is as far from the truth as the divide between the very richest and the very poorest people on this planet.
The truth is that population control is the process by which Global Europe (whites, Caucasians, Aryans) seeks to guarantee its perpetual domination of the rest of the human race because of its own fear of annihilation. According to Dr. Frances Cress-Welsing, it is this fear based on the fact of their numerical minority status and their low level of surface melanin, which drives them to commit the most atrocious crimes against humanity, in particular, the most feared nation of all, Global Africa (Black people).
Is it true that the world is over crowded and moving towards an unsustainable population level?
No. Absolutely not. Overcrowding can be measured by one method only that is whether there are too many people to fit in the space available. The most densely populated continent area in the world is Europe, (see GAP News #7, Population Figures), but do Europeans think there are too many people in Europe? Of course not. But they believe there are too many African and Asian people in Europe. That is not overcrowding that is racism.
What about all those starving Africans? If they can’t feed themselves surely, there must be too many of them.
No, that is not the case. Those “starving Africans”, Asians and other “Third World” peoples produce most of the world’s surplus food. Most of the food they produce are luxury or raw, unprocessed goods which are sold cheaply as exports and re-imported as expensive processed foods.
The main reason though, why there appears to be not enough food to go around is not because the so-called third world cannot feed itself, it is because Global Europe, less than 25% of the world’s population uses or wastes over 80% of the worlds food goods (consumables) but produces less than 15% of it. So the “third world” make up 75% of the world’s population, produce 85% of the world’s consumables and consume less than 20% of all that is consumed. If they consumed as much as they produced, Global Europe would be dying of starvation, not Africa.
Is the African population expanding too rapidly?
Let’s look at the evidence: After being systematically depopulated for 400 years, Africa is now the least populated continent in the world with a density one-sixth of Europe’s. Africa’s death rate is more than twice that of Europe. To be level pegging, Africa’s death rate should also be one-sixth of Europe’s. When these dishonest people talk about population they make reference only to birth rate. They show that Africa’s birth rate is nearly three times that of the European rate, but forget to mention that the infant mortality rate is 5 times higher in Africa.
They never talk about density except in reference to Asia or to say that “Rwanda is the most densely populated country in Africa”. They forget to say it was a quarter the density of any country in Europe. They forget also, to tell you that in order for Africa to get to the same population density as Europe (is Europe overpopulated?) the African birth-rate has to be more than 12 times that of Europe (6 times if the death rate becomes equal) for a whole generation.
So, when they talk about “equalizing” or reducing the African birth rate, while at the same time nurturing conflict, manufacturing famine, and importing disease to increase the death rate further, you begin to get the picture. If the birth rates were made equal and everything else remained the same as they are now, each time Europe’s population doubled Africa’s population would be halved. The world’s population may become “stabilized” as they like to say it, but the percentage ratio between the nations would continue changing to their advantage. (See GAP News #5)
It is understandable then, why Cardinal Alfonso Lopez Trujillo, a senior Vatican official cried that if the precepts of the UN Population Control Conference in Cairo were to be implemented the world would experience “the most disastrous massacre in history”. He should know, it was his organization, the Roman Catholic church, which sanctified the trade in African lives, resulting in the death of over 200 million people.
Some of the liars say that deaths in war time make very little difference to the population growth because after a war birth rates usually increase to compensate. Certainly, that is true when mostly male soldiers are killed. But when two thirds of the female population are murdered, like the Rwandan slaughter, it would take 4 or 5 generations to get back to where it was before the war. And that is the key. The women.
Global Europe have done everything they could to destroy our people but we are still here and still strong. They are now trying, through an apparently limitless line of African and Asian female mercenaries, posing as leaders, to co-opt us. To convince us that regardless of our particular environmental conditions, contrary to our own community’s social and economic needs, it would be in our individual interests to have fewer or no children at all.
Women have the power to determine the fertility or sterility of our nation. It is imperative that we do not allow ourselves to be misled into committing generational suicide. We carry the future of our nation in our hands. We are here because those before us gave us life. Let us give life to our children. We deserve to live.

FRAGMENT:
ABORTION FOR EUGENICS: CONSPIRACY OR SIMPLE CONSEQUENCE?
How one answers the question whether abortion is a tool of racial, gender, or disability eugenics depends very much on how the question is asked. Is legalized abortion a eugenicist conspiracy — a deliberate plot on the part of those favoring abortion rights to reduce the number of people of a given race, sex, or disability? Surely not. At the very least, such motivations form no part of the modern argument for abortion rights. Does unrestricted legal abortion-choice produce a disparate impact resulting in disproportionate numbers of abortions ending the lives of minority, female, and disabled fetuses? Undeniably. The aborted are disproportionately Black, female, and disabled. Is the right to abortion sometimes used, by those exercising the abortion-choice, for eugenics purposes — specifically for the purpose of aborting on the basis of race, sex, or disability? Unquestionably. Some — but not all — of the abortion–disparate impact is attributable to intentional decisions to abort based on a trait of the baby that otherwise would be born.
These are three different questions. Justice Thomas’s concurrence in Box keeps them distinct. Murray’s article, in attempting to critique Thomas, tends to smush these separate questions together in a mildly confusing way.
Begin with Justice Thomas’s Box concurrence itself. Thomas’s opinion compiles an impressive and rightly disturbing narrative of evidence that family planning and abortion advocates in the past embraced the desirability of abortion as an instrument for achieving racial eugenics and for culling persons with disabilities from the population. (There appears to be no evidence that early abortion advocates ever favored abortion for gender-eugenics purposes — aborting girls because they are girls.18×18. This is probably most simply explained by the fact that the technology for discerning the fetus’s sex before birth was not readily available until relatively recently. See, e.g., Juan Stocker & Lorraine Evens, Fetal Sex Determination by Ultrasound, 50 OBSTETRICS & GYNECOLOGY 462, 465 (1977).
Han Chinese academics in Xinjiang in recent years have blamed the high birth rate among the Uyghurs and Kazaks for fostering religious extremism and poverty. According to Zenz’s research, government and academic papers have referred to the birth rate of ethnic minorities in the region as “excessive” and have claimed that the population growth and concentration of ethnic minorities in Xinjiang “weakens national identity and identification with the Chinese Nation-Race (Zhonghua Minzu).”
Population Research Institute
I’ve been meaning to put this together for this a long time now, but we owe it to An0maly that I arrived to finish it, he tipped me over with this great brand new video, where he kills it in his own terms. I just felt I need to round it up and bring more depth and definition that he can’t possibly achieve in his format. The guy is one of the clearest minds on Internet right now.
MORE References
- Black, E. 2003 The Horrifying American roots of Nazi Eugenics, Truth11, George Mason University, Center for History and New Media. http://truth11.com/2009/07/10/the-horrifying-american-roots-of-nazi-eugenics/
- Centers for Disease Control (CDC). 2010. Protective effect of sickle cell trait against malaria-associated mortality and morbidity. http://www.cdc.gov/malaria/about/biology/sickle_cell.html
- Grant, G.1995. Killer angel. Franklin, TN: Ars Vitae Press [Google Scholar]
- Harris, J.2007. Enhancing evolution: The ethical case for making better people. Princeton: Princeton University Press [Google Scholar]
- International Down Syndrome Coalition for Life (IDSC). 2009. Down syndrome: Facts, stats, concern of eugenics. http://www.physiciansforlife.org/content/view/1301/26
- Lev, O., Wilfond B.S., and McBride C.M.. 2013. Enhancing children against unhealthy behaviors-an ethical and policy assessment of using a nicotine vaccine. Public Health Ethics 6: 197–206 [PMC free article] [PubMed] [Google Scholar]
- Lewens, T.2009. Enhancement and human nature: The case of Sandel. Journal of Medical Ethics 35: 354–6 [PubMed] [Google Scholar]
- Porter, R., ed. 2006. The Cambridge illustrated history of medicine, 326 Cambridge, UK: The Press Syndicate of the University of Cambridge [Google Scholar]
- Robertson, J.A.1994. Children of choice. Princeton: Princeton University Press [Google Scholar]
- Savulescu, J., and Kahane G.. 2009. The moral obligation to create children with the best chance of the best life. Bioethics 23(5): 271–90 [PubMed] [Google Scholar]
- Steinbock, B.2002. Sex selection: Not obviously wrong. Hastings Center Report 32: 23–8 [PubMed] [Google Scholar]
- United States Holocaust Museum. 2013. The biological state: Nazi racial hygiene, 1933–1939. http://www.ushmm.org/wlc/en/article.php?ModuleId=10007057
- Willke, J.C.1998. Assisted suicide and euthanasia, 6–8 Cincinnati, OH: Hayes Publishing Co [Google Scholar]
To be continued?
Our work and existence, as media and people, is funded solely by our most generous supporters. But we’re not really covering our costs so far, and we’re in dire needs to upgrade our equipment, especially for video production.
Help SILVIEW.media survive and grow, please donate here, anything helps. Thank you!
! Articles can always be subject of later editing as a way of perfecting them